TITLE
Title:
Persistent burnout theory of chronic fatigue syndrome
Author:
David Jameson
Email: davidj at mind-body-health dot net
Copyright
(c) 2004 David Jameson.
NOTE: there is now a revised and updated version of this paper
ABSTRACT
Chronic
fatigue syndrome (CFS, also known as myalgic encephalomyelitis or ME) is a
severe illness of unknown etiology that has numerous physical, neurological,
physiological and psychiatric symptoms. Many theories have been put forward to
explain the illness, but none has been universally agreed on. This is mainly
due to a failure of existing theories to explain all aspects of the illness,
and the confusing array of symptoms and abnormal findings associated with the
illness, many of which are only seen in a subset of patients.
Certain findings, however, do seem to be central to the illness:
-
Hypothalamic-pituitary-adrenal (HPA) axis disturbances, including a central under-activation of the HPA axis and a loss of circadian rhythm.
-
Abnormalities in the physical structure and activation of the prefrontal cortex (PFC).
-
Psychosocial treatments, the placebo effect, and rehabilitation programmes have been shown to be effective in curing the illness.
Taken
together, these findings point to a possible etiology of the illness. It is
known that the PFC is an important modulator of the HPA axis. The PFC is also
the main brain region involved in the placebo effect and has functions relating
to long-term goals and motivation.
This
hypothesis suggests that CFS is a functional illness caused by structural
changes in the PFC due to long-term morbid stress (or burnout), reinforced by
the illness itself, which can be reversed by psycho-social rehabilitation
treatments.
An outline
of an experiment is presented to examine the correlation between CFS symptoms,
prefrontal cortex activity and morphology, and HPA axis state for patients at
different stages of treatment.
MeSH
Keywords: “Fatigue Syndrome, Chronic”, “Burnout, Professional”, “Stress”, “Hypothalamo-Hypophyseal
System”, “Placebo Effect”, “Prefrontal Cortex”.
INTRODUCTION
Chronic
fatigue syndrome (CFS, also known as ME) is a severe illness of unknown
etiology that has numerous physical, physiological and psychiatric symptoms.
Common triggers for the illness include viral infections – and herpesvirus
family infections in particular – as well as stress (1,2).
Different
studies give widely varying values for the incidence of the illness, but the
general consensus is that it affects between 100 and 300 people per 100,000
population (3). This suggests that CFS is as prevalent as conditions such as
autism, multiple sclerosis or Parkinson's disease, yet the research funding for
CFS is much lower than for any of these other conditions (4).
PREVAILING HYPOTHESES
The main
hypotheses which attempt to explain CFS are: [1] the cognitive behavioural
theory, which proposes that CFS is due to abnormal illness beliefs (5); and [2]
various viral theories, which propose that CFS is caused by a persistent but
as-yet unidentified viral infection. These two theories also represent the
differing viewpoints about the illness: namely, whether it is psychiatric or
organic in origin.
The problem
with [1] is that it does not adequately explain the physical and physiological
symptoms of the illness, such as abnormal immune and HPA axis function.
With [2],
the problem is that no single virus has been shown to infect all CFS patients.
It is more likely that the persistent herpesvirus and other infections seen in
CFS patients are simply a symptom of the suppressed immune function seen in
CFS, rather than being the cause of CFS itself. Also, the viral theories do not
adequately explain why personality and mental attitude have been shown to be
important in determining which patients contract CFS and which recover the most
quickly, and why stress is a significant trigger in initiating the illness
(2,6,7,8).
Psychosocial
treatments do result in recovery from CFS (8,9,10), but the nature of the treatment
itself is important. Cognitive behavioural therapy (CBT) has been shown by some
studies to be effective and by others to be ineffective, and this has been
taken by some researchers as evidence that psychosocial treatments in general
do not work for CFS (11). However, it should be borne in mind that the
patient’s motivation and belief in the treatment are likely to be important to
recovery, and the fact that many CFS patients have a lack of confidence and
motivation in CBT probably has a lot to do with CBT’s lack of effectiveness
(12).
HPA AXIS
The HPA axis
controls the release of cortisol from the outer cortex of the adrenal glands,
via the hypothalamus and the pituitary. Plasma cortisol levels follow a
circadian pattern, with high levels in the morning and lower levels in the
evening. Under conditions of stress the activation of the HPA axis increases,
resulting in a higher cortisol peak.
CFS patients
tend to have a reduced HPA axis response to stress (13), a less pronounced
circadian activation pattern of the HPA axis (14), an overall lower HPA axis
activation (15), and a subset of patients have significantly low cortisol and
shrunken adrenal glands (15,16). Research shows that the patients who recover
most tend to be those who show the greatest improvement in circadian HPA axis
activation (14).
The HPA axis
is commonly thought of as the body’s response to stress, but research over
recent years has shown that it is more accurate to think of it as the body’s
energy expenditure regulator. As well as being involved in the stress response,
the HPA axis also plays a central role in many normal non-stress functions such
as digestion and the activation of the immune system and the autonomic nervous
system (ANS).
PREFRONTAL CORTEX
The
prefrontal cortex is the most evolved part of the brain, and is involved (among
other tasks) in goal-directed activity, long-term planning and motivation. The
prefrontal cortex is also known to be an important modulator of the HPA axis
and is thought to be central to depression – a common CFS symptom – due to
its regulation of neurotransmitters such as serotonin.
Studies have
shown that CFS patients have reduced grey matter and an abnormal distribution
of acetyl-L-carnitine in the bilateral prefrontal cortex (17,18).
A quantitative
electroencephalography (EEG) study of depressed patients has shown that those
patients who respond to a placebo treatment show a significant increase in
prefrontal activity, with no significant change in any other brain region (19).
BURNOUT
Burnout is a
syndrome characterised by symptoms such as fatigue, depression, anxiety,
headaches, disturbed sleep and susceptibility to illness. Research has shown
that a physiological indicator of burnout is reduced HPA axis function and
consequent low plasma cortisol (20,21). As both burnout and CFS show the same
symptoms and physiological profile, as well as the same psychosocial causes, it
is likely that they are in fact the same state, and that CFS is simply a more
severe form of burnout.
PATIENT RECOVERY
While the
various symptoms and triggers of CFS may appear confusing, the reports of
patients who have recovered from the illness all share a common factor. Whether
the patient recovers due to an alternative therapy, a change in lifestyle, or
due to the placebo effect, in each case the common factor is the motivation and
focus provided by treatment.
The most
effective treatments appear to be rehabilitation programmes which fit in with
the patient’s mindset and which encourage the patient to re-integrate back into
normal life while reducing negative stressors and increasing motivating,
enjoyable activities. Anti-depressants can also be useful, especially where
depression and anxiety are present.
HYPOTHESIS
CFS shares
many features with burnout: the symptoms are identical except in severity, the
triggers are the same, and the same abnormalities in HPA axis function are
observed in both. The main reason why burnout has not been considered as a
cause of CFS is twofold: [1] the incorrect notion that burnout is a purely
psychological state of not enjoying one's job; and [2] the fact that CFS
persists even in the absence of stress, when the popular misconception is that
burnout equates to excessive stress.
I postulate
that CFS is caused by a persistent, chronic functional suppression of the HPA
axis, which is essentially the same state as "burnout". This burnout
persists long after the triggering HPA axis stressors have been removed, due to
factors such as negative mental attitude and loss of motivation and perceived
goals, either due to the illness itself or due to other factors.
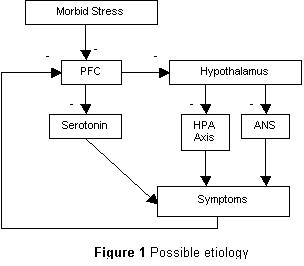
The
prefrontal cortex is likely to be central to the etiology of CFS, as it has
been shown to be abnormal in CFS patients. Also, its function as a modulator of
the HPA axis and its control over serotonin would explain most, if not all, of
the physiological and psychiatric symptoms of CFS. The prefrontal cortex is
also the area of the brain involved in the placebo effect, which is relevant
because many reports of recovery from CFS involve treatments that are
essentially placebos (homeopathy, kinesiology, anti-candida diets, etc.)
The
prefrontal cortex may have a stress protection function, reducing HPA axis
activation and serotonin production during periods of long-term morbid stress.
This results in burnout and depression (or to put it another way, a reduction
in both physical and mental energy and motivation), aiming to force the person
to change the situation. If the situation is not changed, or if the burnout
itself becomes a negative stressor, then the burnout will persist and lead to
CFS.
PROPOSED EXPERIMENT
In order to
further elucidate the etiology of CFS, an experiment is proposed which will
investigate the correlation of prefrontal cortex activity and morphology, HPA
axis function, symptoms and treatment. Such an experiment would give us a
clearer understanding of the nature of CFS, bridging the gap between the
physical and psychiatric debate about the illness.
The
participants would be divided into three groups: [1] a control group of
non-patients; [2] a control group of CFS patients receiving no treatment; and
[3] a group of CFS patients undergoing a rehabilitation programme.
If funding
permits, the following extra groups could be added:
·
A placebo
group of CFS patients receiving a sham drug treatment.
·
An active
treatment group of CFS patients receiving an antidepressant medication.
·
A group of
CFS patients receiving homeopathy treatment.
·
A group of
patients suffering from burnout syndrome, for comparison with CFS.
Prior to the
experiment, participants would be assessed by the Maslach Burnout Inventory to
determine the degree to which the psychological burnout factors are present.
The
participants would be monitored for brain activation (using either QEEG cordance
or MRI scans), brain morphology (using voxel based MRI) and HPA axis activation
(measuring cortisol after awakening and circadian variation in cortisol). At
various stages of the experiment the patients would be assessed on visual
analogue scales to determine subjective recovery from CFS symptoms, with
follow-ups afterwards.
If the
predictions of this theory are correct then patients receiving all treatments
should show changes in the prefrontal cortex and in HPA axis function. Those
patients on the rehabilitation programme should show the most significant
improvement in symptoms as well as changes in prefrontal cortex and HPA axis
activity, and this improvement is more likely to be permanent that in the
placebo group. CFS symptoms are likely to be correlated with the state of the
prefrontal cortex and HPA axis.
REFERENCES
(1) CMO's
CFS/ME Working Group Report, section 3.3.2. Available from: URL: http://www.dh.gov.uk/PublicationsAndStatistics/Publications/PublicationsPolicyAndGuidance/PublicationsPolicyAndGuidanceArticle/fs/en?CONTENT_ID=4064840&chk=D8nces
(2) McLean
SA, Clauw DJ. Predicting chronic symptoms after an acute “stressor”—lessons
learned from 3 medical conditions. Med Hypotheses. 2004;63(4):653-8.
(3) CDC
demographic information about CFS. Available from: URL: http://www.cdc.gov/ncidod/diseases/cfs/demographics.htm
(4) ME
Association Research & Scientific Bulletin, Spring 2003;10:3-4.
(5) Butler
S, Chalder T, Wessely S. Cognitive behavioural therapy in chronic fatigue
syndrome. J Neurol Neurosurg Psychiatry 1991;54:153-158.
(6) White C,
Schweitzer R. The role of personality in the development and perpetuation of
chronic fatigue syndrome. J Psychosom Res 2000 Jun;48(6):515-24.
(7) Creswell
C, Chalder T. Defensive coping styles in chronic fatigue syndrome. J Psychosom
Res 2001 Oct;51(4):607-10.
(8) Szyndler
JE, Towns S, Hoffman RC, Bennett DL. Clinical assessment, management and
outcomes of a group of adolescents presenting with complex medico-psychosocial
conditions. Ann Acad Med Singapore 2003 Jan;32(1):51-7.
(9) Patel
MX, Smith DG, Chalder T, Wessely S. Chronic fatigue syndrome in children: a
cross sectional study. Arch Dis Child. 2003 Oct;88(10):894-8.
(10) Viner R, Gergorowski A, Wine C, Bladen M,
Fisher D, Miller M, El Neil S. Outpatient rehabilitative treatment of chronic
fatigue syndrome (CFS/ME). Arch Dis Child. 2004 Jul;89(7):615-9.
(11) Spence
VA, Abbot NV. Cognitive behaviour therapy for chronic fatigue syndrome. Lancet
2001 Jul 21;358(9277):239-40; discussion 240-1. Comment on: Lancet. 2001 Mar
17;357(9259):841-7.
(12)
Bazelmans E, Prins JB, Hoogveld S, Bleijenberg G. Manual-based cognitive
behaviour therapy for chronic fatigue syndrome: therapists’ adherence and
perceptions. Cogn Behav Ther. 2004;33(3):143-50.
(13) Gaab J,
Huster D, Peisen R, Engert V, Heitz V, Schad T, et al.
Hypothalamic-pituitary-adrenal axis reactivity in chronic fatigue syndrome and
health under psychological, physiological, and pharmacological stimulation.
Psychosom Med 2002 Nov-Dec;64(6):951-62.
(14) MacHale
SM, Cavanagh JT, Bennie J, Carroll S, Goodwin GM, Lawrie SM. Diurnal variation
of adrenocortical activity in chronic fatigue syndrome. Neurophychobiology 1998
Nov;38(4):213-7.
(15) Parker
AJ, Wessely S, Cleare AJ. The neuroendocrinology of chronic fatigue syndrome
and fibromyalgia. Psychol Med 2001 Nov;31(8):1331-45.
(16) Scott
LV, Teh J, Reznek R, Martin A, Sohaib A, Dinan TG. Small adrenal glands in
chronic fatigue syndrome: a preliminary computer tomography study.
Psychoneuroendocrinology 1999 Oct;24(7):759-68.
(17)
Okada T, Tanaka M, Kuratsune H, Watanabe Y, Sadato N. Mechanisms underlying
fatigue: a voxel-based morphometric study of chronic fatigue syndrome. BMC
Neurol. 2004 Oct 4;4(1):14.
(18)
Kuratsune H, Yamaguti K, Lindh G, Evengard B, Hagberg G, Matsumura K, Iwase
M, Onoe H, Takahashi M, Machii T,
Kanakura Y, Kitani T, Langstrom B, Watanabe Y. Brain regions involved in
fatigue sensation: reduced acetylcarnitine uptake in the brain. Neuroimage 2002
Nov;17(3):1256-65.
(19) Leuchter
AF, Cook IA, Witte EA, Morgan M, Abrams A. Changes in brain function of
depressed subjects during treatment with placebo. Am J Psychiatry 2002
Jan;159(1):122-9.
(20) Morgan
CA 3rd, Cho T, Hazlett G, Coric V, Morgan J. The impact of burnout on human physiology
and on operational performance: a prospective study of soldiers enrolled in the
combat diver qualification course. Yale J Biol Med 2002 Jul-Aug;75(4):199-205.
(21)
Pruessner JC, Hellhammer DH, Kirschbaum C. Burnout, perceived stress, and
cortisol responses to awakening. Psychosom Med 1999 Mar-Apr;61(2):197-204.